Chemistry: Polymers, Plastics, and Materials Science
Exploring polymer structure, properties, and uses
Exploring polymer structure, properties, and uses
Chemistry - Grade 9-12
- 1
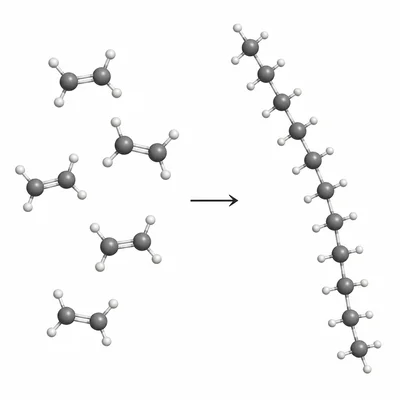
Define the terms monomer and polymer. Use polyethylene as an example in your explanation.
- 2
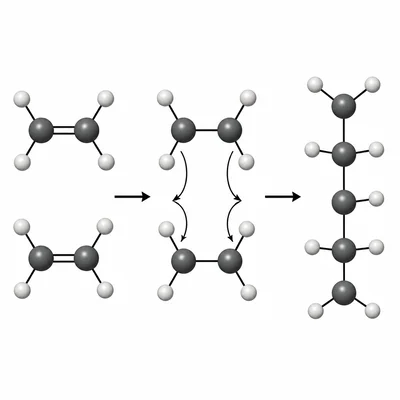
Ethene has the formula C2H4 and can form polyethylene through addition polymerization. Describe what happens to the carbon-carbon double bond during this process.
- 3
A polymer sample has an average molar mass of 84,000 g/mol. Its repeating unit has a molar mass of 28 g/mol. Calculate the average degree of polymerization.
- 4
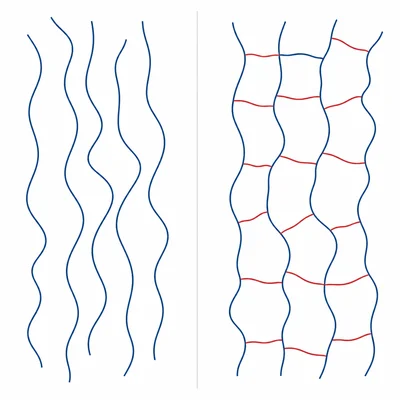
Compare thermoplastics and thermosetting plastics. Include one property difference and one example use for each type.
- 5
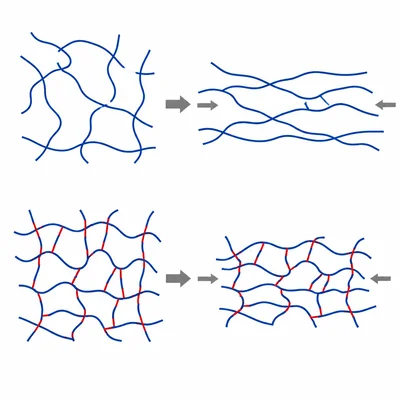
Explain how cross-linking affects a polymer's flexibility, melting behavior, and strength.
- 6
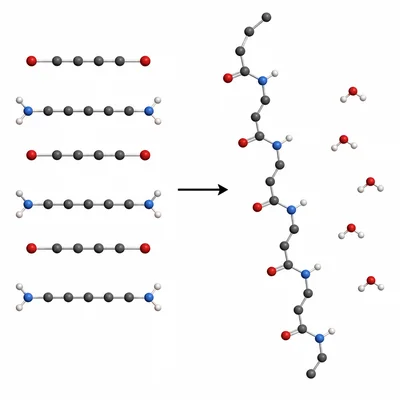
Nylon is formed when a diamine reacts with a dicarboxylic acid, releasing small molecules such as water. Identify this as addition polymerization or condensation polymerization and explain your choice.
- 7
A plastic labeled PET is commonly used in beverage bottles. Describe one useful property of PET for this application and one environmental concern related to PET bottles.
- 8
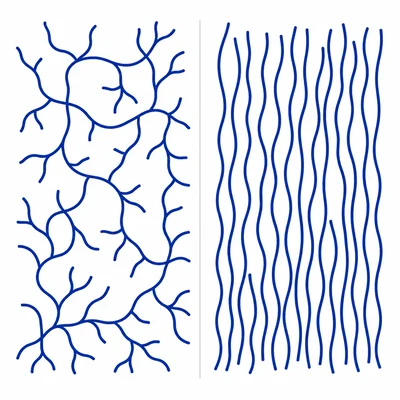
Explain why low-density polyethylene, LDPE, is usually more flexible than high-density polyethylene, HDPE.
- 9
A material scientist wants a plastic for a reusable food container that can handle repeated washing, moderate heat, and bending without cracking. List two polymer properties that should be tested and explain why each matters.
- 10
Explain the difference between biodegradable plastics and recyclable plastics.
- 11
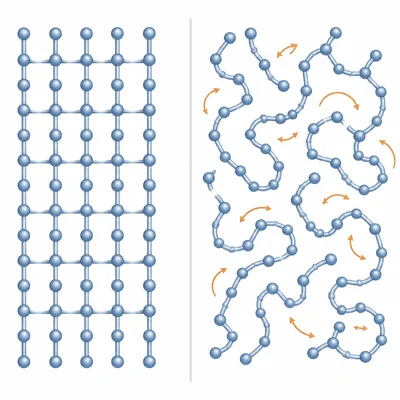
A polymer has a glass transition temperature, Tg, of 80 degrees Celsius. Describe how the material is likely to behave at 25 degrees Celsius and at 100 degrees Celsius.
- 12
A recycling facility receives a mixed batch of plastics containing PET, HDPE, PVC, and polystyrene. Explain why sorting plastics by type is important before recycling.
Related Tools
Related Labs
Related Infographics
Related Cheat Sheets
More Chemistry Worksheets
States of Matter
Grade 2-3 · 8 problems
Science: Atoms Elements and the Periodic Table
Grade 6-8 · 15 problems
Science: Chemical Reactions and Balancing Equations
Grade 6-8 · 15 problems
Science: Properties of Matter
Grade 2-3 · 15 problems
More Grade 9-12 Worksheets
Linear Equations
Math · 8 problems
Cell Biology
Biology · 8 problems
Reading Comprehension
Language Arts · 8 problems
Historical Thinking & Evidence
Social Studies · 8 problems