Chemistry
Titration Curves
Titration Curves
Related Tools
Related Labs
Related Worksheets
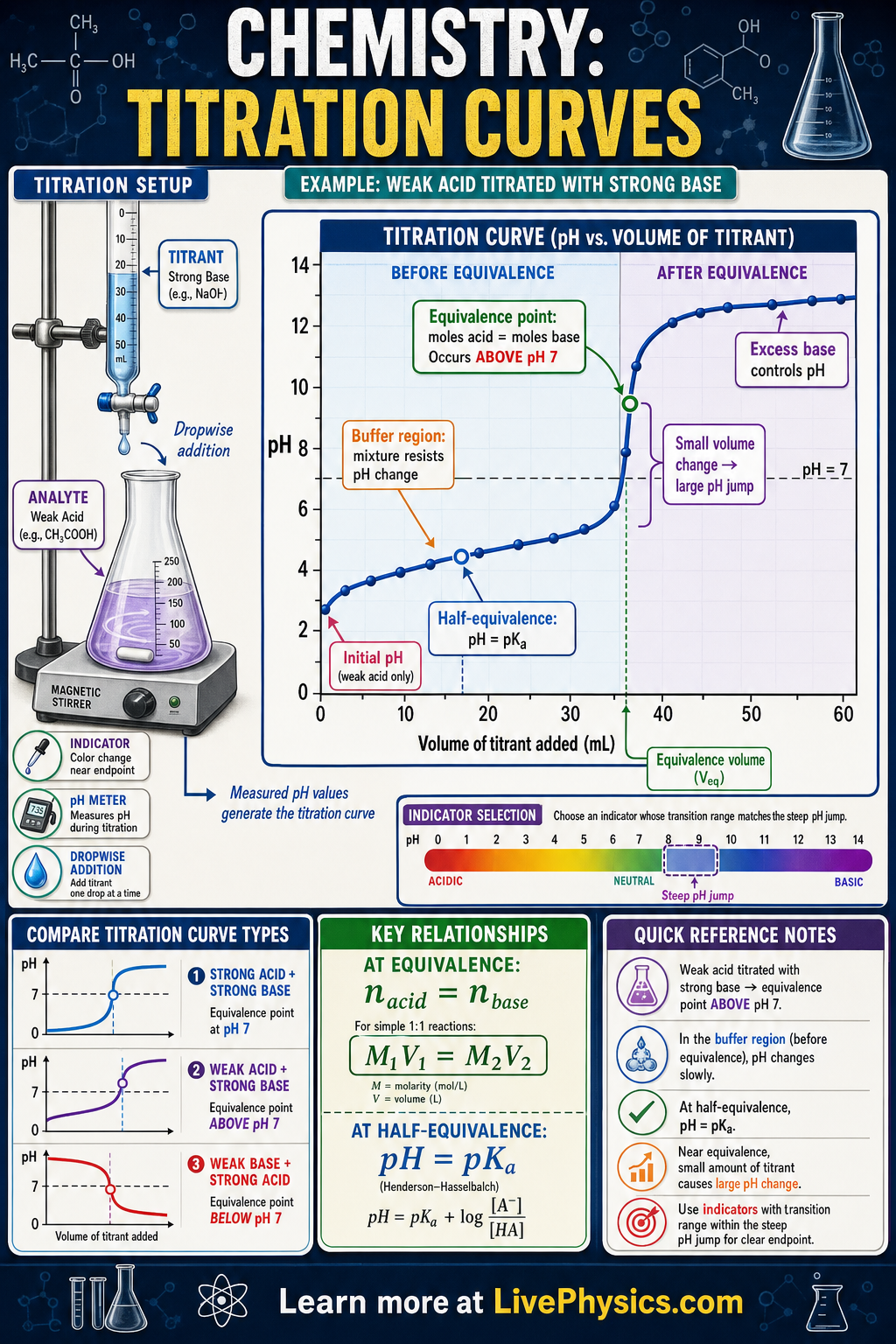
A titration curve shows how the pH of a solution changes as a titrant is added from a burette. It is one of the most useful graphs in acid-base chemistry because it reveals the strength of the acid or base, the equivalence point, and the best indicator to use. The curve is usually steep near the equivalence point, where a small added volume causes a large pH change. Reading these curves helps chemists measure unknown concentrations accurately.
Key Facts
- At the equivalence point, moles of acid and base have reacted in the exact stoichiometric ratio.
- For a strong acid and strong base titration, the equivalence point is at pH = 7 at 25 °C.
- Moles of solute = M x V, where M is molarity and V is volume in liters.
- For a monoprotic acid titrated with a strong base, MaVa = MbVb at the equivalence point.
- At the half-equivalence point of a weak acid titration, pH = pKa.
- A good indicator changes color within the steep vertical region near the equivalence point.
Vocabulary
- Titration
- A lab method used to determine an unknown concentration by reacting it with a solution of known concentration.
- Titrant
- The solution of known concentration that is added from the burette during a titration.
- Analyte
- The solution being tested, usually placed in the flask before the titration begins.
- Equivalence point
- The point where the amount of titrant added has completely reacted with the analyte according to the balanced equation.
- Buffer region
- A part of a weak acid or weak base titration curve where pH changes slowly because both a weak species and its conjugate are present.
Common Mistakes to Avoid
- Confusing the endpoint with the equivalence point: the endpoint is when the indicator changes color, while the equivalence point is the stoichiometric completion of the reaction.
- Using milliliters directly in mole calculations: volume must be converted to liters when using moles = M x V.
- Assuming every equivalence point has pH = 7: weak acid and weak base titrations have equivalence points above or below 7 because conjugate ions affect pH.
- Choosing an indicator only by color: the indicator must change color over the steep pH range near the equivalence point to give an accurate result.
Practice Questions
- 1 A 25.0 mL sample of HCl is titrated with 0.100 M NaOH. The equivalence point occurs at 32.5 mL of NaOH. What is the molarity of the HCl?
- 2 A weak acid is titrated with NaOH. The half-equivalence point occurs after 18.0 mL of NaOH has been added, and the pH is 4.76. What is the pKa of the acid, and at what volume is the equivalence point?
- 3 A titration curve starts at low pH, has a buffer region, and reaches an equivalence point above pH 7. Identify the type of titration and explain why the equivalence point is basic.